|
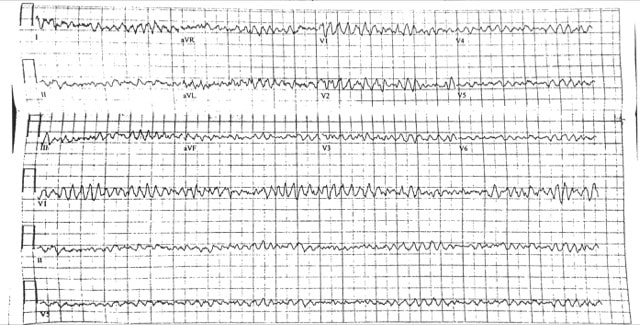
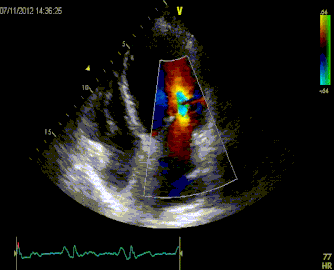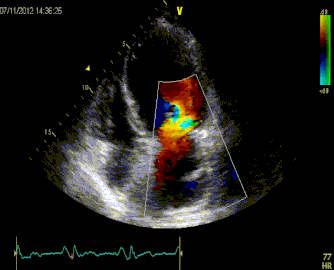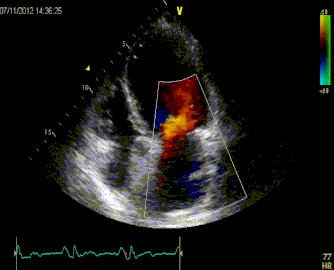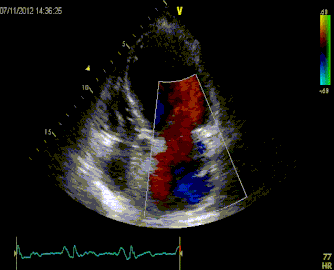
45 M presents to the ED with lightheadedness after his LVAD “Low Flow” alarm sounded. He has a normal mental status, and MAP ~ 65 mmhg What is the rhythm?
Ventricular Fibrillation. There are no P waves nor organized QRS complexes. This distinction between coarse and fine V Fib is irrelevant. In theory fine V Fib indicates it has been a longer duration but management is not different. You may look at the EKG and question whether the patient has Torsades. The initial distinction is irrelevant as the patient is likely unstable / pulseless and needs electricity. Torsades can only be diagnosed with prolonged QT on the patient’s preceding or ensuing EKG. How would you typically manage this rhythm?
ACLS approach to tachycardias:
1.Does the patient have a pulse? 2. Shockable rhythm or not? If Pulse and unstable → Synchronized cardioversion (the electricity is timed to the cardiac cycle). If the patient is pulseless then analyze the rhythm and determine is it “Shockable” (Pulseless VTach or VFib) or “Non-shockable” (PEA or Asystole). The first step is immediate CPR in both scenarios until you can get the defibrillator to defibrillate the shockable rhythms (electricity not timed to cardiac cycle). ****THESE ARE THE PATIENTS THAT ACLS SAVES**** How would you manage this rhythm in lvad patients?
Call for help when they hit the door. You should NOT perform CPR in an LVAD patient unless you can confirm they are in cardiac arrest. Most often the pump needs troubleshooting. Arrhythmias are not uncommon. You can attempt chemical cardioversion (amiodarone) or electrical cardioversion. Pads do NOT go over the LVAD. Use the anterior/posterior positions.
Take Home Points 1. It is not important to distinguish between types of VFib → the answer is defibrillation 2. Torsades de Pointes can only be diagnosed if the preceding/ensuing EKG has prolonged QT 3. Do NOT perform CPR on LVADs unless Cardiac Arrest confirmed (eg MAP 0 mmHg) 4. When performing defibrillation or cardioversion in LVAD patients place pads Anterior / Posterior Resources: Partyka C and Taylor B. Review article: ventricular assist devices in the emergency department. Emerg Med Australas. 2014 Apr; 26(2): 104-12. Link MS, Berkow LC, Kudenchuk PJ et al. Part 7: Adult Advanced Cardiovascular Life Support: 2015 American Heart Association Guidelines Update for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2015; 132:S444
0 Comments
Massive Pulmonary Embolism
Submassive Pulmonary Embolism
ManagementIn patients with severe hypoxia and shortness of breath, consider NIPPV early, even before CT diagnosis. As the patient has increased Right sided failure and RV dilation, the patient will continue to have worsening RV ischemia and decreased RV contractility. As RV afterload increases you begin having worsening right sided failure and worsening cardiac output. In patients who require inotropes, consider norepi and dobutamine.
Systemic ThrombolyticsThe use of systemic thrombolytics are supported by most protocols. Many RTCs have evaluated the use of thrombolytics, including half-dose and catheter-directed TPA. While mortality improves with thromolytic use in massive PEs, RTCs evaluating lytics in submassive PEs have been mixed. Catheter-directed intervention is a safe and effective treatment for both submassive and massive PEs.
61 year old presents to the ED with chest pain radiating to his shoulder What is the diagnosis?
Why Not...
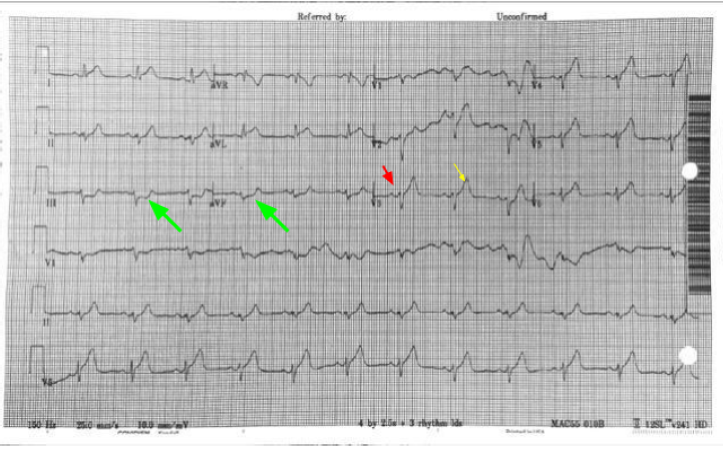
Why is this not Benign Early Repolarization?
The differentiation of benign ST elevations (BER) and Anterior STEMI can be difficult. There are a number of characteristics which are suggestive of BER vs. STEMI. Make sure to examine for other ischemic changes. Our job is to exclude MI and BER should be diagnosis of exclusion. Some features suggestive of each in below table. Learn more
Consider using the Subtle Anterior STEMI calculator in cases where you are concerned, but the EKG may be difficult to decipher Since October 20th, 2017, an increased number of mumps cases in college students have been reported from DC Universities. Mumps is an acute viral disease caused by a paramyxovirus. Symptoms include fever and swelling of unilateral or bilateral parotid or other salivary glands. Swelling usually peaks in 1 to 3 days, and can last 2 or more days before subsiding. In approximately one-third of cases, mumps infection may present with nonspecific or primarily respiratory symptoms, and can also be subclinical. A person with mumps is generally contagious two days before onset of symptoms to 5 days after. The incubation period is 14-18 days from the time of exposure, but can be as long as 25 days. Mumps spreads from person to person via respiratory secretions or saliva or through fomites. Diagnostic Testing Please obtain the following specimens as soon as mumps is suspected: · Oral or buccal swab samples. Massage the parotid gland for 30 seconds and store in 2 ml of viral transport medium. Refrigerate samples at 4°C for shipment within 24 hours and ship on cold packs (store at -70°C if delayed). · Serum in a red-top or serum separator tube for IgG and IgM testing Detailed specimen collection instructions can be found on the CDC website (https://www.cdc.gov/mumps/lab/specimen-collect.html). It is critical to perform mumps PCR for confirming the diagnosis as serology test result can be misleading especially in previously vaccinated persons. For additional questions regarding specimen collection, please email doh.epi@dc.gov. Reporting Cases to DOH Suspected cases of mumps infection should be reported IMMEDIATELY to the Department of Health at (202)-442-9371 or via the DC Reporting and Surveillance Center (DCRC) on our Infectious Disease website (https://doh.dc.gov/service/infectious-diseases). Reports should be made at time of initial clinical suspicion of mumps infection. If diagnosis of mumps is considered and diagnostic testing are ordered, the case should be reported at that time. Do not wait for laboratory confirmation. Vaccination Updated guidelines from the Advisory Committee on Immunization Practices (ACIP) indicate that a 3rd dose can be recommended in outbreak settings for identified high-risk groups. Healthcare providers can consult with DC DOH to determine whether an additional vaccine dose is indicated. A fact sheet on (https://doh.dc.gov/page/disease-fact-sheets). Please contact the DC DOH Division of Epidemiology–Disease Surveillance and Investigation at: Phone: 202-442-8141(8:15am-4:45pm) | 844-493-2652 (after-hours calls) Fax: 202-442-8060 | Email: doh.epi@dc.gov
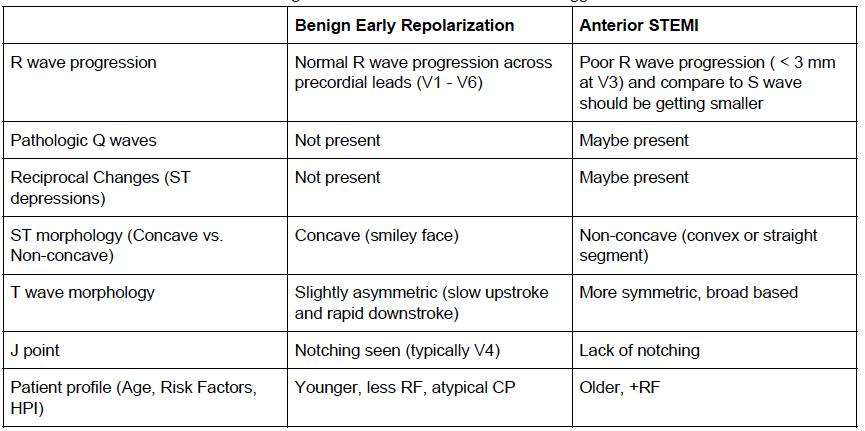
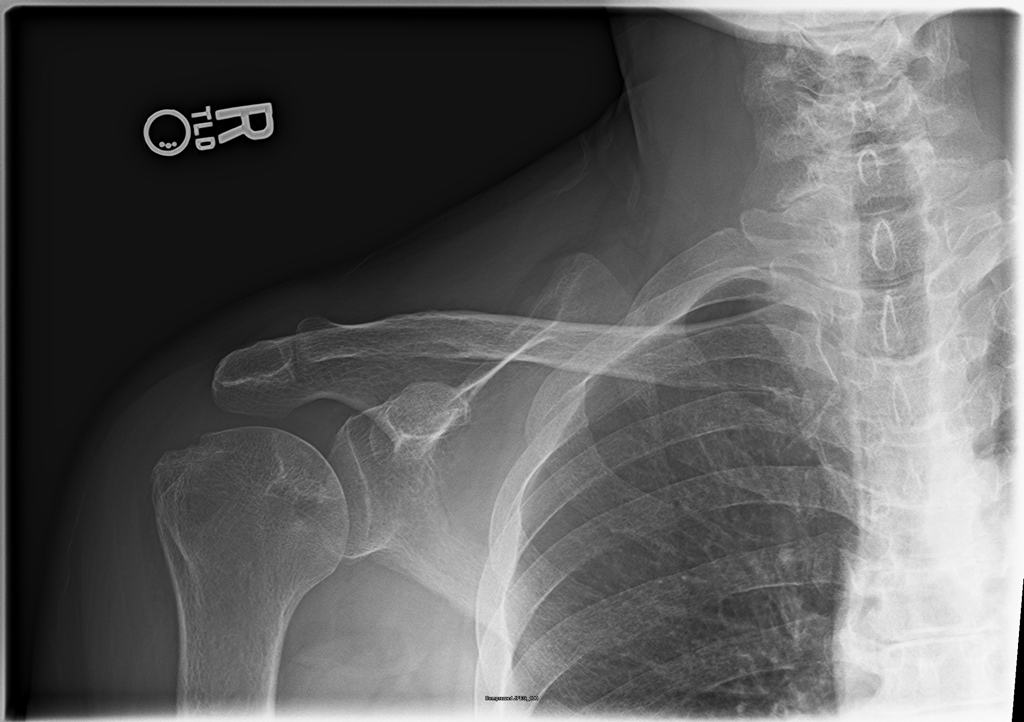
57 yo female with a history of hypertension, osteoarthritis, and asthma with chronic steroid use, presenting with multiple complaints, including chronic right shoulder pain that has been worsening the past week. The pain is constant and worse with any ROM of the right shoulder. Physical exam, including Spurling's Test, is otherwise unremarkable. On further discussion, the patient has had a remote hip replacement for avascular necrosis, and no recent traumas.
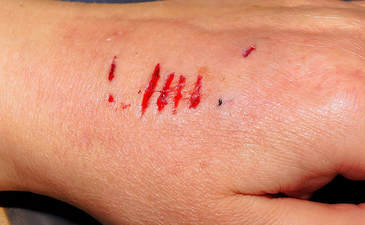
Dog bites have commonly mixed flora, with a 50% incidence of pasturella. Primary closure can be done most places except the hands and feet. For open wounds (such as the hands and feet) patients should receive prophylactic augmentin to cover most bacteria, similar to cat or human bites. Dog bites that are closed typically do not require prophylactic antibiotics. Wound CareThere is little evidence-based medicine to support a specific fluid for wound care. Normal saline or tap water have both been shown to have a similar infection rate (4-6%). Betadine-containing solutions have not been shown to improve outcomes or decrease infection rates. Some surgical research supports the use of betadine solutions. ClosureThe primary reason for wound closure is for improved cosmesis. A RCT of patients presenting with dog bites found that closure of dog bite wounds does not increase the risk of infection or complication, and closed wounds had better cosmetic appearance. Hand wounds, regardless of treatment, did have a higher rate of infection. AntibioticsPatients presenting with hand wounds from dog bites should receive prophylactic antibiotics. Per a Cochrane review, there is some evidence showing that dog bites in the hand have reduced infection rates with prophylactic antibiotics. The same review did not show a reduction in cats or for all dog bites.
Mechanical Complications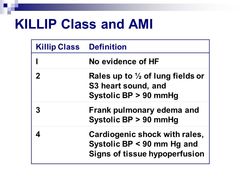 Treatment for mechanical post-MI complications includes vasodilators and ACE inhibitors, as well as blood thinners in cases which have thrombi.
Ischemic Complications
Pericarditis
Suicidal behavior, both attempts and ideation, are a common ER complaint, accounting for 4% of all visits. One million people in the US attempt suicide each year, and 1.2% of all US deaths are due to suicide. As the holidays are coming soon, I felt this would be a good time to review how we can ensure a safe discharge for those who may not meet inpatient criteria. TL;DR: ER interventions can decrease suicide attempts by 30%. Use the below dotphrase to improve your documentation in patients who are being discharged. A study published this July in JAMA Psychiatry found the implementing an ER intervention can prevent future suicidal behavior, with a number needed to treat between 13 and 22. The study utilized a primary (universal) and a secondary screening tool administered by the nurse and provider, followed by discharge resources and a follow-up phone call. The first step is similar to the current triage safety questions, asking patients about suicidal thoughts or attempts. Those who screen positive are identified and a secondary screening form (PDF) was completed by the physician. This form uses known risk factors for suicide to determine if there should be a psychiatric consultation (plan for suicide, intent, prior inpt treatment, and substance abuse). The third step of their intervention consisted of a safety plan which identified triggers for suicidal thoughts, how to get help, and what followup steps need to happen. All patients received a followup phone call after discharge. The study found that the intervention group had a 30% decrease in total suicide attempts than those receiving typical treatment. The study is interesting, but YOU DON’T NEED THESE FORMS TO HELP SAVE LIVES. Most of us are probably already incorporating most of this into our treatment of those patients being discharged with outpatient followup!
After initial evaluation, the patient endorses _ depressive symptoms. The patient has _ prior suicide attempts, currently _ abusing medications or drugs, and _ family history of suicide. The patient has _ judgement as evidenced by _, and displays _ insight into their symptoms. Although the patient is _ not currently endorsing active suicidal or homicidal ideation, and appears to have a good plan for follow up, I will consult psychiatry to provide outpatient resources and ensure a safe discharge plan prior to disposition.
One-third of Americans self report as obese, and bariatric surgery has become a leading treatment option. Approximately 200,000 procedures are performed annually, most commonly gastric sleeves.
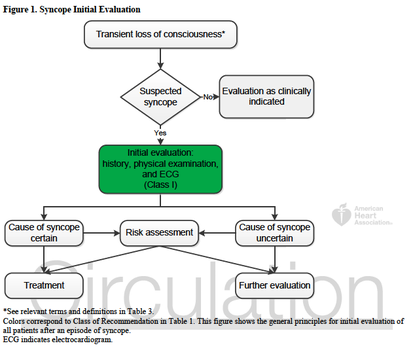
In-line with the month’s cardiology theme, this note from the TR desk will discuss the evaluation of syncope, and the brand new (2 days young) ACCF/AHA Guidelines for the Evaluation and Management of Patients With Syncope. TL;DR: Watch Sonal’s syncope lightning rounds, and consider using the San Fran or Boston (both links) syncope rules. Use the FREE DOTPHRASE to help remind yourself of syncope patients who may required admission. The 2017 recommendations are the most evidence-based guidelines yet for the evaluation of syncope, and based on an extensive literature review and committee discussion which included emergency medicine physicians, ACEP, and SAEM! Studies report an estimated prevalence of a single episode of syncope at 19% of the population. Females have a higher prevalence of syncope, and those with chronic cardiac and vascular issues are more likely to have recurrent syncope. The most common cause of syncope is unknown in most cases (37%), followed by Reflex Syncope (21%), Cardiac Syncope (9%) and Orthostatic Hypotension (9%). The recommendations include a relatively worthless chart (above) showing how to initially evaluate a patient with syncope, recommending a detailed history and examination including orthostatics and evaluation for new murmurs, and a neurological exam. There is also Level B evidence that syncope patients should be evaluated with an EKG (This is a level A recommendation from ACEP). Given Cardiac Syncope has a high risk of recurrence and death, this seems appropriate. The article goes on to recommend targeted lab testing in any patient where the initial evaluation and diagnosis is not crystal clear. If you were here for Dr. Batra’s excellent discussion on syncope last year, none of this is surprising to you. The recommendations also discuss the use of many of the current Syncope Risk Scores, including the San Francisco Syncope Rule, Boston Syncope Rule, and Del Rosso (All three of these have Negative Predictive Values of 99%). This is probably the most important part, as we need to determine which patients are safe to discharge, versus those who should be admitted. When looking across all three rules, a few bit picture items come out; abnormal EKGs, a history of CHF or heart disease, age >65, and syncope either without a prodrome or while supine, are all concerning for higher-risk Cardiac Syncope. Additionally, we should consider alternative causes of syncope, including pulmonary embolism (DO NOT bring up the PESIT Trial), pregnancy, anemia/GI bleeding, and neurologic causes. As such, you can use the dotphrase below to help remind yourself of these potential causes, and reasons to consider admission for monitoring, Holter monitor placement, and potentially further workup. Dotphrase/MacroPt here with syncope/near syncope. Seizure less likely given history and exam. No neurologic deficits on exam indicating a stroke, and no signs of head trauma or injury. Given no signs of trauma or neurologic deficit, I will withhold further imaging of the head per ACEP Choosing Wisely Recommendations. Additionally, ACEP clinical policy on syncope evaluation recommends laboratory testing and advanced investigative testing such as echocardiography or cranial CT scanning need not be routinely performed unless guided by specific findings in the history or physical examination.
Patient’s history includes _ prior syncopal events, _ CAD, _ DVT/PE, _ seizures. Cardiac evaluation today shows _ murmur, _ JVD, _ peripheral pulses and _ lower extremity edema. EKG today _ without signs of Brugada or QT shortening or prolongation. _ PE risk factors. Neurologically _. Blood glucose _, _signs of hypoxia during event or currently, and _ intoxication complicating the patient’s presentation. Given this, the patient’s episode _. Risk Factors for Serious Cause: older age, pre-syncopal exertion, history of cardiac disease including heart failure, family history of sudden death, recurrent episodes, recumbent episode, prolonged loss of consciousness, chest pain or palpitations. Age >65, and Hct <30% San Francisco Syncope Rule (.ekmdmsanfran)CHF History Hct <30% EKG Abnormality SOB SBP < 90 mmHg at triage |
Categories
Archive
February 2018
Please read our Terms of Use.
|
||||||||||||||||||